Gasoline Blending – NMR Application Overview
This application marketed by NMR Process Systems LLC – Paper Presented at Experimental NMR Conference – 1996
Much has been written on the subject of NIR and Mid-IR spectroscopy applied to gasoline blending. Over the past 15 years repeated attempts have been made to validate these on-line systems. Bottom line is the systems validate when close attention is paid to the modeling effort. The modeling effort very quickly becomes cumbersome owing to the large number of models that must be maintained. As an example – for octane, separate models must be developed for high and low octane, for mid octane, for CARB fuel, for oxygenated fuel (EtOH, MTBE, TAME), for finished blends, for each blend component, for European fuels, etc. Dozens of models are required just for RON. Continuous model updates are required after each crude slate change (the analyzer is not available until the new gasolines have been produced and placed in the model. Bluntly, if onsite expertise is not continuously available the project will fail and the analyzer will never be validated.
NMR is quite different on the modeling side of the gasoline blending project. Being a relatively insensitive technique (by insensitive we mean that no small component change can have a dramatic effect on the spectrum) the NMR does not have the continual model maintenance issue of spectral changes caused by crude slate changes. The NMR is also an inherently linear spectroscopy across all hydrocarbon types and compositions (this is not the case with IR or NIR). This inherent linearity means that one can build a single RON model for example that span 70 octane to 110 octane and have an SECV of 0.9 octane numbers (mainly caused by the repeatability/reproducibility of the engine test at the high and low ends of the model range). Narrowing the range will yield more accurate models with SEP’s of 0.2 to 0.5 octane numbers.
Another advantage of NMR over other spectroscopies is the orthogonality of the chemical types within the NMR spectrum itself. Polynuclear aromatics (di, and tri), mono-aromatics, internal olefins, terminal olefins, oxygenates, xylenic, toluenic, substituted aromatics, CH, naphthenes, CH2 and CH3 all fall into their own distinct chemical shift regions allowing easy modeling of all chemical based parameters (PIONA, Polynuclear aromatics, benzene, xylenes, toluene, FIA, total aromatics, oxygenates, etc.).
Distillation and density are readily modeled. More difficult is RVP which does need several models to be able to accurately predict RVP of different gasoline types.
Below are a few figures that show the data rich nature of the NMR spectrum. With a little know-how it is possible to be able to look at an NMR spectrum and estimate the RON, aromatic content or any other parameter. The spectral differences are huge.
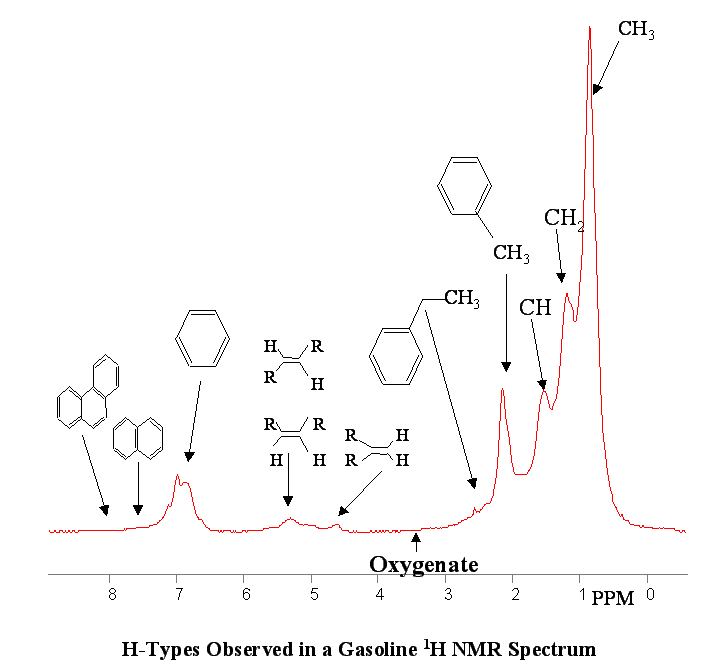
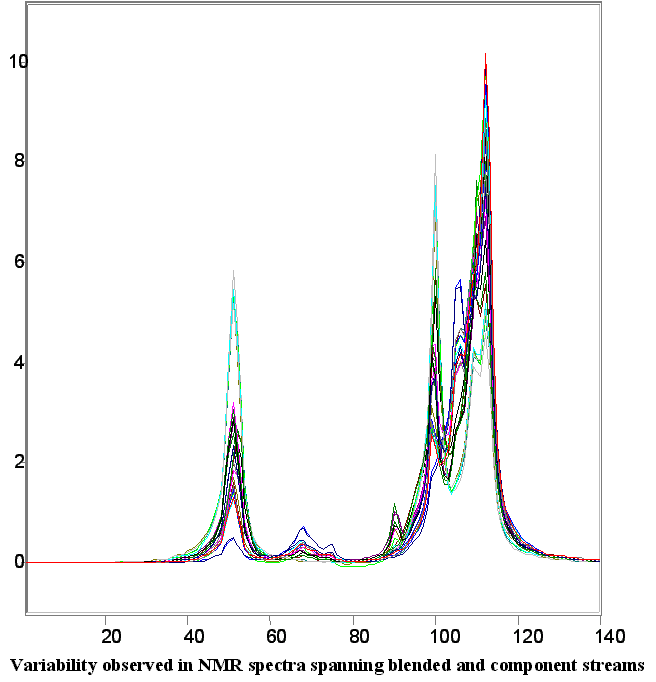
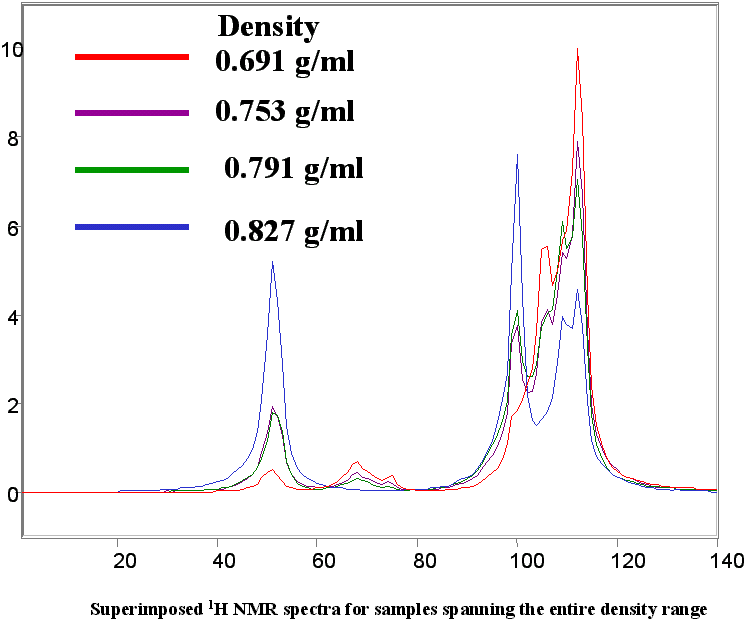
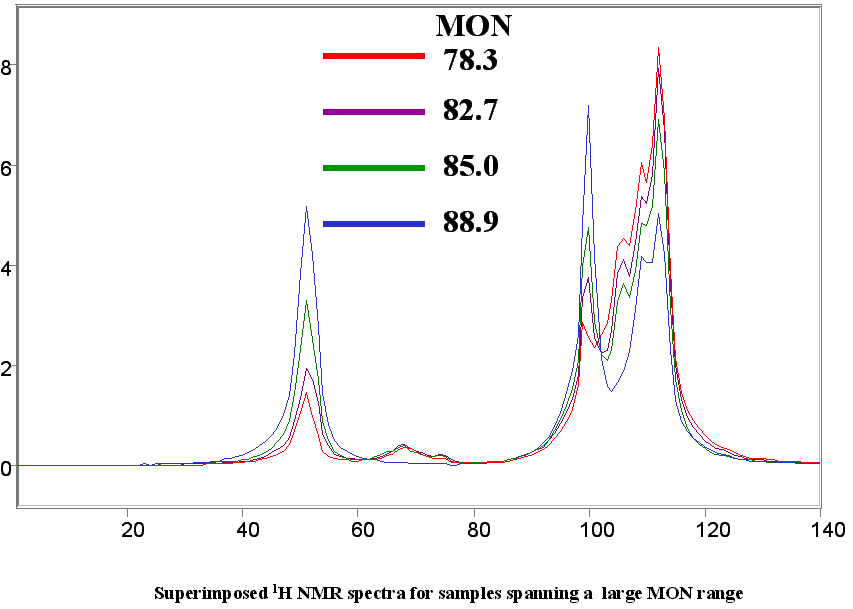
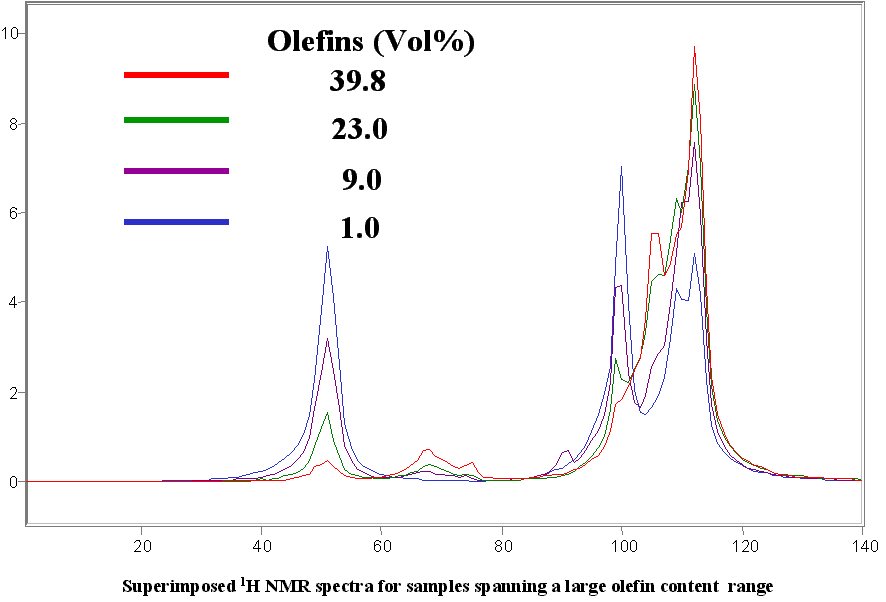
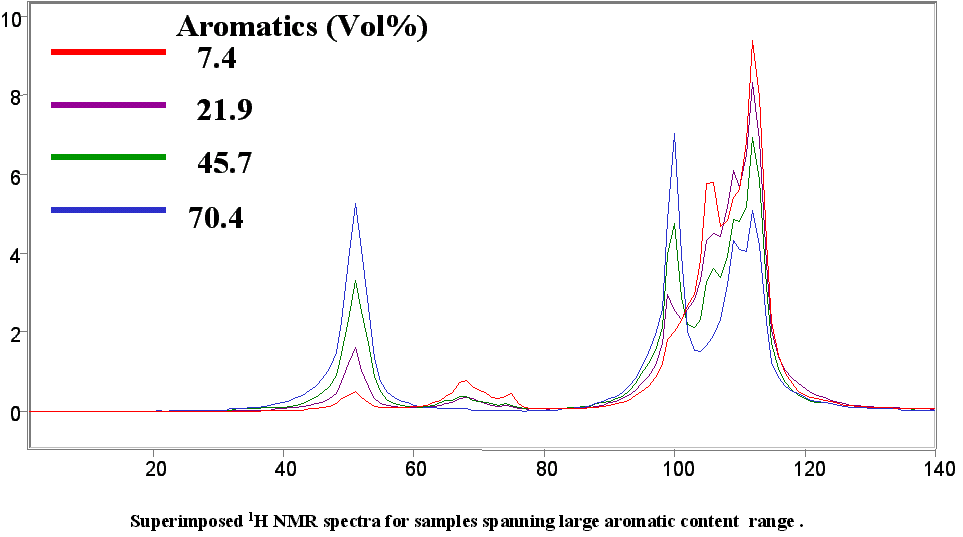
Below is a listing of ASTM repeatabilities and reproducibilities. If the NMR system is validated following the procedures set out in ASTM D3764 (or D6122) the the expected agreement between the NMR predicted number and the average value from multiple lab tests on the same sample will approach the ASTM repeatability for the various tests. If the NMR is validated against single point lab data (one sample, one test) then the expected agreement between lab and NMR will be closer to the ASTM reproducibility for the various test methods.
ASTM Precision (Gasoline Testing)
- API – ASTM D4052
| SG | Repeatability | Reproducibility |
| 0.68 – 0.97 | 0.0001 | 0.0005 |
NMR Correlation cannot attain reproducibility level of density test. NMR Reproducibility will be within 0.0025 g/litre
- RVP – ASTM D5191
| Parameter | Repeatability | Reproducibility |
| Dry Vapor Pressure Equivalent (DVPE) | 0.00807 (DVPE+B) | 0.0161 (DVPE +B) |
B=124 kPa or 18.0 psi depending on the units of DVPE
- FIA – ASTM D1319
| FIA Precision, Oxygenate Free Sample | ||||||
| Level, Volume % | Repeatability | Reproducibility | ||||
| Aromatic | Olefin | Saturate | Aromatic | Olefin | Saturate | |
| 1 | 0.4 | 0.3 | 1.7 | 1.1 | ||
| 5 | 0.7 | 0.9 | 0.8 | 1.5 | 3.7 | 2.4 |
| 15 | 1.2 | 1.5 | 1.2 | 2.5 | 6.1 | 4.0 |
| 25 | 1.4 | 1.8 | 1.5 | 3.0 | 7.4 | 4.8 |
| 35 | 1.5 | 2.0 | 1.7 | 3.3 | 8.2 | 5.3 |
| 45 | 1.6 | 2.0 | 1.7 | 3.5 | 8.5 | 5.6 |
| 55 | 1.6 | 2.0 | 1.7 | 3.5 | 8.5 | 5.6 |
| FIA Precision, Oxygenate Containing Sample | |||
| Range | Repeatability | Reproducibility | |
| Aromatic | 13 – 40 | 1.3 | 3.7 |
| Olefin | 4 – 83 | 0.2578X0.6 | 0.8185X0.6 |
| Saturate | 45 – 68 | 1.5 | 4.2 |
X = the volume percent of olefins
- Distillation – ASTM D86 (Group 1)
| Evaporated Point | Manual | Automated | ||
| Repeatability | Reproducibility | Repeatability | Reproducibility | |
| IBP | 6 | 10 | 7 | 13 |
| 5% | r1 + 1.2 | R1 + 2.0 | r2 + 1.8 | R2 + 3.2 |
| 10% | r1 | R1 | r2 + 1.8 | R2 + 1.3 |
| 20% | r1 | R1 | r2 | R2 + 1.3 |
| 30 – 70% | r1 | R1 | r2 | R2 |
| 80% | r1 | R1 | r2 | R2 – 1.7 |
| 90% | r1 | R1 – 2.2 | r2 | R2 – 3.5 |
| 95% | r1 | R1 – 1.7 | r2 + 1.8 | R2 |
| FBP | 7 | 13 | 8 | 16 |
r1 = 0.864 (DF/DV%) + 2.186 R1 = 1.736 (DF/DV%) + 3.589 r2 = 0.673 (DF/DV%) + 2.036 R2 = 1.998 (DF/DV%) + 4.711
DF/DV% = slope at evaporated point, °F/Volume%
- Benzene – ASTM D3606
| Component | Range, Volume % | Repeatability | Reproducibility |
| Benzene | 0.1 – 1.5 | 0.03X + 0.01 | 0.13X + 0.5 |
| Benzene | >1.5 | 0.03 | 0.28X |
X = the mean volume % of the component
- Oxygenates – ASTM D5599
Repeatability and Reproducibility Vary Depending on Oxygenate Component
| Component | Repeatability | Reproducibility |
| Ethanol | 0.03 (X0.49) | 0.27 (X0.80) |
| MTBE | 0.05 (X0.58) | 0.10 (X0.95) |
| ETBE | 0.04 (X0.86) | 0.25 (X0.79) |
| TAME | 0.04 (X0.58) | 0.24 (X0.69) |
X= Mean Vol% of Component
- Aromatics – ASTM D5769
| Component | Repeatability | Reproducibility |
| Total Aromatics | 0.027(X+4.4) | 0.10(X+4.4) |
X= Mean Vol% of Component
- RON – ASTM D2699
| Range | Repeatability | Reproducibility |
| 80 | Unknown | 1.2 |
| 85 | Unknown | 0.9 |
| 90 | 0.2 | 0.7 |
| 95 | 0.2 | 0.6 |
| 100 | Unknown | 0.7 |
| 105 | Unknown | 1.1 |
| 110 | Unknown | 2.3 |
- MON – ASTM D2700
| Range | Repeatability | Reproducibility |
| 80 | Unknown | 1.2 |
| 85 | 0.3 | 0.9 |
| 90 | 0.3 | 1.1 |
| 95 | Unknown | 1.1 |
| 99 | Unknown | 1.5 |
| 100 | Unknown | 1.1 |
| 105 | Unknown | 1.8 |
For more information on this topic please contact us.